

|
Article
Daniella Silva Oggiam1
Juliana Vallim Jorgetto2
Guilherme Luiz Chinini3
Denise Miyuki Kusahara4
Mônica Antar Gamba5
1 ![]() 0000-0001-9814-8924. Universidade Federal de São Paulo, Brazil. prof.daniella.oggiam@unipinhal.edu.br
0000-0001-9814-8924. Universidade Federal de São Paulo, Brazil. prof.daniella.oggiam@unipinhal.edu.br
2 ![]() 0000-0003-3758-5201. Universidade Federal de São Paulo, Brazil. juliana.vallim@unifesp.br
0000-0003-3758-5201. Universidade Federal de São Paulo, Brazil. juliana.vallim@unifesp.br
3 ![]() 0000-0001-8548-0696. UniPinhal, Brazil. prof.guilherme.chinini@unipinhal.edu.br
0000-0001-8548-0696. UniPinhal, Brazil. prof.guilherme.chinini@unipinhal.edu.br
4 ![]() 0000-0002-9498-0868. Universidade Federal de São Paulo, Brazil. dkusahara@unifesp.br
0000-0002-9498-0868. Universidade Federal de São Paulo, Brazil. dkusahara@unifesp.br
5 ![]() 0000-0003-1470-4474.
Universidade Federal de São Paulo, Brazil. antar.gamba@unifesp.br
0000-0003-1470-4474.
Universidade Federal de São Paulo, Brazil. antar.gamba@unifesp.br
* This article is part of the doctoral dissertation "Effects of Monochromatic Infrared Light Intervention on Neuropathic Pain Levels in People with Diabetes Mellitus: A Randomized Controlled Clinical Trial," Nursing Graduate Program, Universidade Federal de São Paulo, Brazil. It was financed by the Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP), grant number 2017/26282-3.
** El presente artículo hace parte de la tesis doctoral "Efectos de la intervención con luz infrarroja monocromática en los niveles de dolor neuropático de personas con diabetes mellitus: ensayo clínico controlado aleatorizado", Programa de Posgrado en Enfermería, Universidade Federal de São Paulo, Brasil. El proyecto contó con la financiación de la Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP), subvención número 2017/26282-3.
*** El presente artículo hace parte de la tesis doctoral "Efectos de la intervención con luz infrarroja monocromática en los niveles de dolor neuropático de personas con diabetes mellitus: ensayo clínico controlado aleatorizado", Programa de Posgrado en Enfermería, Universidade Federal de São Paulo, Brasil. El proyecto contó con la financiación de la Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP), subvención número 2017/26282-3.
Received:
26/10/2020
Sent to peers: 01/12/2020
Approved by peers: 10/06/2021
Accepted: 16/07/2021
To reference this article / Para citar este artículo / Para citar este artigo: Oggiam DS, Jorgetto JV, Chinini GL, Kusahara DM, Gamba MA. Distal Symmetric Polyneuropathy Pain in Diabetes Mellitus. Aquichan. 2021;21(3):e213X. DOI: https://doi.org/10.5294/aqui.2021.21.3.7
Theme: Chronic care and health promotion
Contribution to the discipline: Neuropathic pain is one of the most disabling complications in patients with diabetes mellitus. Systematically screening neuropathic pain through instruments validated for this purpose and recognizing factors associated with it enable primary health care nurses to establish early interventions for pain prevention and management.
ABSTRACT
Objective: To evaluate
neuropathic pain (NP), its intensity, and complications in people with type 2
diabetes mellitus (T2DM) in a city of eastern São Paulo.
Method: Cross-sectional
study conducted with 96 individuals with T2DM served by primary health units in São João da
Boa Vista-SP. The following instruments were used to screen NP: Michigan
Neuropathy Screening Instrument, Leeds Assessment of Neuropathic Symptoms and Signs, Douleur Neuropathique 4, and
Brief Pain Inventory. The data were analyzed using descriptive and inferential
statistics, with a 5 % significance level.
Results: Of the 96 people
with T2DM for longer than five years, 22.9 % had pain. NP was related to high
levels of fasting blood glucose (mean = 214 ± 65.58 mg/dl; p = 0.0002), glycated hemoglobin (mean = 8.8 ± 0.11 %; p < 0.001),
absence of a balanced diet (p = 0.0066), obesity (p = 0.023), and high blood
pressure (p < 0.001).
Conclusion: Higher values of glycated hemoglobin rates increased three times the chance
of NP. The screening and management of painful diabetic neuropathy is a
challenge but adopting a screening protocol supports the secondary prevention
of this manifestation.
KEYWORDS (Fonte DeCS): Diabetic neuropathy; chronic pain; diabetes mellitus; complications; physiotherapy; physical therapy specialty; nursing.
RESUMEN
Objetivo: evaluar el dolor neuropático (DN), su intensidad y sus complicaciones en
personas con diabetes mellitus tipo 2 (DM2) en una ciudad del este de São Paulo.
Método: estudio transversal
realizado con 96 individuos con DM2 atendidos en unidades
básicas de salud en São João da Boa Vista-SP. Se utilizaron los
siguientes instrumentos para rastrear el DN: el instrumento para la detección
de la neuropatía de Michigan, la evaluación de signos y síntomas neuropáticos de Leeds, el cuestionario Douleur Neuropathique 4 y el inventario breve
del dolor. Los datos se analizaron mediante estadística descriptiva e
inferencial, con un nivel de significancia del 5 %.
Resultados: de las 96 personas con DM2
durante más de cinco años, el 22,9
% tenía dolor. El DN se relacionó con
niveles altos de glucosa en sangre en ayunas (media = 214 ± 65,58 mg/dl; p =
0,0002), la hemoglobina glucosilada (media =
8,8 ± 0,11 %; p < 0,001), la ausencia de una
dieta (p = 0,0066), la obesidad (p = 0,023) y la hipertensión
arterial (p < 0,001).
Conclusión: los altos valores en
las tasas de hemoglobina glucosilada aumentaron tres veces la probabilidad de DN. La detección y el tratamiento de
la neuropatía diabética dolorosa es un desafío, pero la adopción de un
protocolo de detección contribuye a la prevención secundaria de esta
manifestación.
PALABRAS CLAVE (Fuente DeCS): Neuropatías diabéticas; dolor crónico; diabetes mellitus; complicaciones; fisioterapia; enfermería.
RESUMO
Objetivo: avaliar a dor neuropática (DN), sua intensidade e suas complicações em pessoas com diabetes mellitus
tipo 2 (DM2) numa cidade do
leste de São Paulo, Brasil.
Materiais e método: estudo transversal realizado com 96 indivíduos com DM2 atendidos em unidades
básicas de saúde em São João da Boa Vista, São Paulo. Foram utilizados os seguintes instrumentos para rastrear a DN: o instrumento para detectar a neuropatia de Michigan, a avaliação de sinais e sintomas neuropáticos de
Leeds, o questionário de Douleur Neuropathique 4 e a escala breve da dor. Os dados foram analisados por meio de estatística descritiva e inferencial, com um nível de significância de 5 %
Resultados: das 96 pessoas com DM2 durante mais de cinco anos, 22,9 % sentiam dor. A DN esteve relacionada com níveis altos de glicose em sangue em jejum (média = 214 ± 65,58 mg/dl; p = 0,0002), hemoglobina glicosilada (média = 8,8 ± 0,11 %; p < 0,001), ausência de dieta (p = 0,0066), obesidade (p = 0,023) e hipertensão arterial (p < 0,001).
Conclusões: os altos valores nas taxas de hemoglobina glicosilada aumentaram três vezes a probabilidade de DN. A detecção e o tratamento da neuropatia diabética dolorosa são um desafio, mas a adoção de um protocolo de detecção contribui para a prevenção secundária dessa manifestação.
PALAVRAS-CHAVE (Fonte DeCS): Neuropatias diabéticas; dor crônica; diabetes mellitus; complicações; fisioterapia; enfermagem.
Introduction
Diabetic peripheral neuropathy (DPN) is prevalent in about 50 % of diabetes mellitus (DM) cases. It is characterized by chronic clinical or subclinical manifestations resulting from injuries to the somatic peripheral nervous system involving nerve fibers. Progressively, DPN becomes a risk factor for developing a series of clinical complications such as degeneration of the nerves in the distal region of the lower limbs, ulcers, amputations, and neuropathic pain (NP) (1-3).
Among DPN cases, 75 % had distal symmetric polyneuropathy (DSP) of multifactorial pathogenesis, caused by a metabolic dysfunction. It results in oxidative stress and inflammation of peripheral nerves, in addition to variations in voltage-dependent sodium channels that worsen DSP and NP (4,5).
NP, the most incapacitating clinical complication of DSP among DPNs, is caused by extensive lesions in fine Aβ-, Aδ-, and C-type nerve fibers that adversely affect the sensory and motor nerve components. It has different clinical manifestations, pathophysiological mechanisms, development, and evolution. NP appears as spontaneous chronic pain, without any stimulus, or painful hypersensitivity triggered by sensitive neuronal injury (6-10). Pathophysiological studies report loss of nerve fibers, axonal degeneration, and demyelination associated with oxidative stress. The latter is induced by chronic hyperglycemia that causes mitochondrial damage and thus neural apoptosis (11, 12).
The prevalence of NP is around 20 % and increases according to DM duration, advancing age, and a lack of glycemic control. The prevalence is likely to rise as the global population ages. Therefore, screening NP is vital for implementing public policies aimed at early intervention and improving the quality of life of those affected. There is a growing need for adequate PDN and NP screening to make diagnosis and clinical treatment easy and effective (2).
Validated assessment instruments in the Portuguese language, which are easy to use and fairly accurate, can assist in screening the signs and symptoms of NP with high predictive values for diabetic neuropathy and NP. These instruments include Michigan Neuropathy Screening Instrument (MNSI) (13, 14), Leeds Assessment of Neuropathic Symptoms and Signs (LANSS) (15), Douleur Neuropathique 4 (DN4) (16), and Brief
Pain Inventory (BPI) (17), which provide better clinical accuracy in DM (3, 18).
The MNSI is widely used in clinical trials worldwide, facilitating the screening of diabetic neuropathy with clinical history and physical examination (13, 14, 19). It comprises 13 items for clinical history: numbness, burning, sensitivity to touch, cramps, tingling, pain, temperature sensitivity, weakness of muscles, night-time worsening of symptoms, and tactile sensitivity. These are associated with 10 items for physical examination of the feet: appearance (deformities, dry skin, calluses, infections, fissures), ulcerations, deep ankle reflex, hallux vibration perception test with 128 Hz tuning fork, and 10 g monofilament test (19,20).
For screening DM NP, the LANSS and DN4 instruments are extensively used due to their high predictive value. The first differentiates NP from nociceptive pain; it consists of seven items with specific pain characteristics associated with physical assessment, allodynia, and pain threshold at needle examination. Its score ranges from 0 to 24 points; less than 12 points characterize nociceptive pain and more than 12 points NP (15,21,22). The DN4 questionnaire has 10 questions, three of which are for the physical examination of signs such as burning, tingling, numbness, changes in temperature sensitivity, and tactile stimulus. The score ranges from 0 to 10; equal to or greater than four indicates NP (23).
BPI is a numerical instrument that assists in assessing pain intensity and location, pain interference in daily life activities, social activities, mood, sleep, and walking. It allows quantifying pain self-perception through a score ranging from 0 to 10 for each item evaluated; above four points indicate significant pain (15,24).
The multidisciplinary team working in primary health care, including nursing and physiotherapy, is essential in the care and guidance of people with DM, as it screens, monitors, and treats complications arising from DM and the lack of adequate glycemic control. These professionals are skilled in using these assessment tools for effective monitoring, patient self-care, and treatment guidance.
Given the need and importance of screening diabetic neuropathy complications with instruments that calculate their prevalence, this study aims to analyze NP through validated instruments, measure its intensity, and identify related complications in a group of individuals with type 2 diabetes mellitus (T2DM) covered by the primary health care network in a city in the state of São Paulo.
Method
This cross-sectional study on NP, its intensity, and complications involved people with T2DM using primary health care in São João da Boa Vista (SP) in 2018.
Data collection and NP screening were carried out from November 2018 to April 2019 using four validated instruments to measure NP and DPN after approval by the Research Ethics Committee (No. 2.303.253). This research guarantees subjects' confidentiality and privacy regarding their personal data and autonomy.
Participants were selected by stratified random sampling from a population of 2,542 individuals diagnosed with T2DM (International Classification of Diseases (ICD) code E10 and E11), registered in the extinct Hiperdia program, in 13 primary health units in the different regions of the municipality.
The study included individuals residing, domiciled, and registered in the municipal public network of São João da Boa Vista, having cognitive conditions to participate in the study (alertness, mental soundness to meet the study requirements voluntarily), aged over 30 years, and with a diagnosis of T2DM for five years or more. Additionally, the following exclusion criteria were adopted: alcoholism, decompensated hypothyroidism, signs of spinal cord compression, stenosis or radiculopathy, chronic pain of another nature such as vascular pain and nociceptive pain, severe peripheral artery disease, ischemic or hemorrhagic stroke associated with T2DM, uncontrolled high blood pressure, pregnancy, lactation, malignant disease in the lower limbs, active systemic cancer, posterior knee and spine surgery, score greater than 12 on the Hamilton Depression Rating Scale (HDRS) (25), no glycemic monitoring, and no glycated hemoglobin in the last six months. The HDRS is used in the assessment and exclusion processes, as depression may interfere with the pain level
We investigated variables related to sociodemographic characteristics, laboratory, clinical aspects, and complications related to T2DM, lifestyle habits, comorbidities, treatments, hospitalization for the disease, treatment satisfaction in primary health units, and assessment of NP and its intensity.
The data of each patient with T2DM were obtained at the health units during an appointment scheduled by telephone by the responsible researcher and from information recorded in the medical records. Participants signed the Informed Consent and filled out a form on sociodemographic characteristics, lifestyle, and clinical history. For screening diabetic neuropathy and NP and measuring pain intensity and complications, we used four validated instruments, namely, the MNSI (13,14) to assess the characteristic symptoms of diabetic neuropathy; the LANSS (15) to differentiate the type of neuropathic or nociceptive pain; the DN4 (16) to identify specific symptoms of NP, and the BPI (17) for the numerical quantification of pain.
Based on instrument results, NP was determined when LANSS scores were greater than or equal to 12, DN4 scores were greater than or equal to 4, and BPI scores were greater than 4. The MNSI classified individuals with DPN in scores above 6 (26) through data computed in a software called Fuzzy, which classifies the evaluated neuropathic symptoms. The HDRS considered a score greater than or equal to 9 points for depression symptoms (25).
Predictive values for the instruments that assessed NP (LANNS, DN4, and BPI) were calculated from the probabilistic expression of an individual with T2DM having a score sufficient to determine NP.
The complications related to the DM studied were assessed by temperature sensitivity test with a test tube and water at different temperatures, light touch test with cotton on the feet for allodynia, and simple yes or no questions on painful symptoms. Sleep quality was evaluated by asking the individual with T2DM whether they had had a peaceful night of sleep without pain or a night disturbed by pain in their feet.
The sample was calculated assuming about 20 % of people with NP of the total number of individuals with T2DM in the municipality. Then, 194 people with T2DM were recruited, approximately 15 individuals per primary health unit. We employed the Simple Random Sampling Calculation to determine the proportion according to Equation (1):
![]()
Where n: sample size; N: population size; Z: abscissa of the normal distribution (1.65 to 90 % confidence level); d: maximum permissible error (decimal); p: the proportion of one of the events (p= 0.5 is allowed when the previous proportion of pilot samples is not known) (27).
The prevalence of NP was calculated as a static measure through specific assessments to identify the number of individuals with T2DM and NP from the total sample. We used Equation (2):
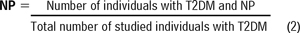
The specificity of the instruments used for NP (LANNS, DN4, and BPI) was calculated using the likelihood ratio (LR) to obtain their predictive values.
The data were analyzed using descriptive and inferential statistics. Quantitative variables were presented as mean, median, and standard deviation, while categorical variables were represented by absolute and relative frequency. After parametric and non-parametric calculations, the variables associated with NP were quantified, building a ROC curve, and defining the odds ratio value. Moreover, the Hosmer-Lemeshow test was employed to identify the model's fit to the data obtained.
The odds ratio was calculated as a measure of the intensity of association based on a multivariate logistic model to quantify the chances of events enabling the onset of NP in the studied population.
Results
A total of 194 medical records of people with T2DM were analyzed. According to the eligibility criteria, 96 people were included in the study, three died before any assessment, and 95 people were excluded. Thirty-four (33.32 %) had less than five years of T2DM, 26 (25.48 %) had spinal disorders with medullary compression, stenosis, or herniated disc; 17 (17.46 %) refused to answer the questionnaire or have their feet examined; 12 (11.76 %) had ischemic stroke, and 6 (11.98 %) severe artery disease, hypothyroidism, decompensated DM, or poliomyelitis.
The general age range of individuals who participated in the study was between 30 and 87 years. Most were female (53; 57.6 %), Caucasian (53; 55.2 %), married (65; 68.2 %), residing in the rural area (69; 72 %), with elementary education (21; 21.9 %), income from agriculture (69; 72.0 %), and classified according to per capita income as class C (46; 47.8 %). Their mean time since T2DM diagnosis was 12 ± 6.5 years, average fasting glucose was 184 ± 12 mg/ dL, and average glycated hemoglobin was 7.8 % ± 0.01.
Diabetic neuropathy and NP were identified in 22 (22.9 %) individuals (95 %; CI: 0.2032-0.3867). Table 1 shows data on sociodemo-graphic characterization and lifestyle habits considered risk factors for comorbidities, with and without diabetic neuropathy and NP.
Table 1. Qualitative variables: Sociodemographic characteristics, risk factors, and comorbidities of individuals with T2DM, according to NP
Variables |
Diabetic neuropathy with no pain n = 74 n (%) |
Diabetic neuropathy with pain n = 22 n (%) |
p-Value |
95 °% CI |
Age range > 60 years |
53 (71.6) |
16 (72.7) |
0.494 |
[0.1323; 0.3315] |
Female |
45 (60.8) |
16 (63.6) |
0.7771 |
[0.1519; 0.3727] |
Caucasian |
43 (58.1) |
11 (50.0) |
0.4614 |
[0.0963; 0.3111] |
Smoking and alcoholism |
28 (37.8) |
12 (54.5) |
< 0.001 |
[-3.0967; 1.9655] |
Physical activity |
22 (29.7) |
01 (4.5) |
0.2502 |
[-1.6242; 0.3756] |
Balanced diet |
29 (39.1) |
03 (13.6) |
0.0066 |
[-3.2911; -0.6223] |
High blood pressure |
64 (86.4) |
21 (95.4) |
< 0.001 |
[0.1554; 0.3387] |
Obesity (BMI > 30) |
31 (41.9) |
15 (68.2) |
0.023 |
[-0.1707; 2.0456] |
Dyslipidemia |
1 (41.8) |
15 (68.1) |
0.4719 |
[0.1906; 0.4615] |
Note. BMI = Body Mass Index. Source: Own elaboration
We identified a statistically significant association between alcoholism and smoking, not having a balanced diet, and comor-bidities such as obesity, high blood pressure, and the peripheral neuropathy with pain. Table 2 presents the results regarding T2DM control in individuals with and without NP.
Table 2. Variables related to clinical history in the control of T2DM, according to NP
Variables |
Diabetic
neuropathy with no pain |
Diabetic
neuropathy with pain |
p-Value |
95 %% CI |
T2DM time - years (mean ± SD ) |
8 ± 6.55 |
12 ± 6.57 |
0.0039 |
[0.0382; 0.1866] |
Fasting blood glucose - mg/dL (mean ± SD ) |
172 ± 55.4 |
214 ± 65.58 |
0.0002 |
[0.0074; 0.0235] |
Glycated hemoglobin - % (mean ± SD) |
7 ± 0.12 |
8.8 ± 0.11 |
< 0.001 |
[59.1416; 63.2348] |
Monitoring frequency of capillary blood glucose - n (%) Daily Weekly Monthly |
18 (24.3) |
4 (18.2) |
0.4093 |
[0.657; 0.979] |
Form of glycemic control - n (%) Oral medication Insulin Both |
34 (45.9) |
2 (9.0) |
0.119 |
[0.869; 1.019] |
Note. SD=standard deviation.
Source: Own elaboration
We identified a statistically significant association between alcoholism and smoking, not having a balanced diet, and comorbidities such as obesity, high blood pressure, and the peripheral neuropathy with pain. Table 2 presents the results regarding T2DM control in individuals with and without NP.
There was a statistically significant difference between individuals with and without NP and disease duration, fasting blood glucose, and glycated hemoglobin. Patients with peripheral neuropathy and pain had longer T2DM duration and higher levels of glycemia and glycated hemoglobin (Table 2).
Table 3 shows the mean and standard deviation of the scores obtained through the tools for assessing diabetic neuropathy and screening NP. It was possible to identify higher scores in people with diabetic neuropathy and NP in relation to the mean scores of each administered instrument.
Table 3. Scores of the instruments for screening painful neuropathy in T2DM, according to NP
Variables |
Diabetic neuropathy with no pain (mean ± SD ) |
Diabetic neuropathy with pain (mean ± SD ) |
p-Value |
MNSI |
3.70 (3.36) |
13.54 (2.75) |
< 0.001 |
LANNS |
3.34 (3.80) |
16.10 (2.55) |
< 0.001 |
DN4 |
1.40 (1.54) |
7.78 (1.89) |
< 0.001 |
BPI |
1.49 (2.09) |
7.67 (1.58) |
< 0.001 |
Note. LANSS: Leeds Assessment of
Neuropathic Symptoms and Signs; BPI: Brief Pain Inventory; DN4: Douleur Neuropathique 4; MNSI:
Michigan Neuropathy Screening Instrument.
Source: Own elaboration
BPI measures pain intensity as mild (up to 2 points), moderate (3 to 7 points), and severe (8 to 10 points). The average score of individuals with T2DM who had NP was 7.67 points, considered as moderate to severe pain intensity.
Additional analysis of the positive predictive value of specific instruments for assessing NP (LANNS and DN4) demonstrated a specificity of 84.85 % and 98.4 %, respectively. Meanwhile, the BPI numerical pain scale, nonspecific to NP, showed a specificity of 81.2 % for this type of pain.
Table 4 shows the most apparent manifestations and complications associated with T2DM and diabetic neuropathy with pain compared to people with diabetic neuropathy without pain.
Table 4. Complications associated with T2DM, according to NP
Variables |
Diabetic neuropathy with no pain n (%) |
Diabetic neuropathy with pain n(%) |
p-Value |
95 %% CI |
Temperature sensitivity changes |
33 (44.5) |
6(27.2) |
0.0021 |
[0.0406; 0.2671] |
Allodynia |
1 (1.3) |
14 (63.6) |
< 0.001 |
[0.80709; 1] |
Night-time worsening of symptoms |
12 (16.2) |
19 (86.3) |
< 0.001 |
[0.4414; 0.7844] |
Poor sleep patterns |
40 (54.0) |
15 (68.2) |
0.0054 |
[0.1550; 0.3904] |
Depression symptoms |
10 (13.5) |
8 (36.3) |
0.0022 |
[0.21488; 0.674] |
Amputation |
1 (1.4) |
3 (13.6) |
0.0176 |
[0.3256; 1] |
Ulcers |
7 (9.46) |
4(18.2) |
0.0137 |
[0.0794; 0.6479] |
Source: Own elaboration
Hospitalization in the last 12 months was not significant for any of the groups, generally around 2.08 %, and there was no difference between them.
Regarding the prevalence of individuals who used specific medications for NP, only one (4.54 %) individual with diabetic neuropathy and pain took pregabalin. The individuals assessed reported 67 % satisfaction with the treatment of the Unified Health System.
Table 5 shows the multivariate analysis of the association between demographic variables, risk factors, T2DM-related factors, and the complications of T2DM with diabetic neuropathy and NP.
Table 5. Association between demographic variables, risk factors, and complications of T2DM with peripheral neuropathy and NP
Variables |
Estimation |
SD |
95 %% CI |
p-Value |
Intercept |
-19.2796 |
8.7772 |
[-42.4236; -5.2077] |
0.0281 |
Fasting blood glucose |
-0.0167 |
0.0138 |
[-0.0481; 0.0086] |
0.2276 |
HB1Ac |
204.8823 |
93.8609 |
[42.7929; 428.4307] |
0.0290 |
DM time |
0.0344 |
0.0888 |
[-0.1493; 0.2322] |
0.6986 |
Male |
-1.6534 |
1.9437 |
[-6.8181; 1.5126] |
0.3950 |
Diet for DM |
-4.0907 |
2.2755 |
[-9.8344; -0.3553] |
0.0722 |
Previous use of tobacco and alcohol |
-0.2400 |
1.8659 |
[-3.9079; 3.9309] |
0.8977 |
Smoking and alcoholism |
-1.5543 |
3.3681 |
[-8.8636; 5.1881] |
0.6445 |
Obesity |
0.7386 |
1.2261 |
[-1.7612; 3.3311] |
0.5469 |
Decreased vibratory sensitivity |
-1.0485 |
1.4033 |
[-4.3624; 1.5688] |
0.4550 |
Absent vibratory sensitivity |
0.2160 |
2.0288 |
[-3.9072; 4.5434] |
0.9152 |
Decreased temperature sensitivity |
3.3169 |
2.0229 |
[0.0725; 8.8623] |
0.1011 |
Temperature sensitivity |
-1.9473 |
2.3561 |
[-6.7377; 3.2267] |
0.4085 |
Physical activity |
4.0447 |
2.1055 |
[0.6459; 9.3609] |
0.0547 |
Night-time worsening of symptoms |
0.7194 |
1.8985 |
[-3.2168; 4.6288] |
0.7048 |
Good sleep quality |
-0.9796 |
1.3649 |
[-4.2455; 1.5637] |
0.4730 |
Amputation |
1.1531 |
1.9445 |
[-2.5738; 5.4062] |
0.5532 |
Allodynia indicator |
0.9914 |
1.5877 |
[-2.1986; 4.3773] |
0.5324 |
Ulcer indicator |
-0.4523 |
1.8685 |
[-4.4390; 3.3579] |
0.8087 |
Age group indicator |
2.2560 |
3.9863 |
[-5.9017; 10.2529] |
0.5714 |
Race indicator |
-1.7476 |
2.0689 |
[-6.1938; 2.2572] |
0.3983 |
Source: Own elaboration
Estimates of the logistic model with multivariate analysis allowed isolating the influence of Hb1AC on diabetic neuropathy with NP (p = 0.0290). For every 1 % of increased Hb1Ac, there was an increase of 2.8954 times the chance of developing this complication. When calculating the area for the ROC curve, we obtained a value of 0.8225, indicating the model's good discrimination (OR 2.8954; -95 %: 2.7519; 3.0465; p < 0.001).
Discussion
Diabetic neuropathy manifested by NP is one of the most devastating and disabling complications of T2DM. Despite its medical and social relevance, there are few studies on its main clinical and epidemiological characteristics, making it even more difficult to screen, assess, and diagnose and impairing adequate therapeutic management.
This research identified diabetic neuropathy and NP in 22.9 % of the individuals. Patients with peripheral neuropathy and pain had T2DM for longer and higher levels of glycemia and glycated hemoglobin. For every 1 % of increased Hb1Ac, there was a 2.8fold increase in the chance of developing this complication. A statistically significant association was identified between the use of alcohol and cigarettes, failure to have a balanced diet, and co-morbidities such as obesity, high blood pressure, and peripheral neuropathy with pain. In addition, people with NP experienced complications such as changes in temperature sensitivity, allodynia, amputations, ulcers, worsening sleep quality, and depression symptoms significantly more frequently than those without pain.
NP assessment and diagnosis in people with T2DM is little targeted and specific concerning their symptomatology. Thus, it is necessary to exclude other causes not to be confused with pains related to diabetes itself, such as symptoms of spinal lesions, which is a widespread disorder (28). About a quarter of the individuals included in this research had NP, which spotlights the magnitude of this complication and the importance of making health professionals aware of the clinical screening of DPN and NP using specific scales for identifying their signs and symptoms (29,30).
We noted a high prevalence coefficient of painful diabetic neuropathy among the sample studied compared to data from other Brazilian regions that have a lower prevalence of NP, such as Santa Catarina with 16.7 % (31). This may result from the association between prolonged illness time and inadequate management of people with diabetes in the region, where there is no standard monitoring and strict treatment in health units. A lack of a multi-disciplinary team responsible for the comprehensive monitoring of these patients leads to inadequate glycemic control and, thus a high frequency of complications inherent to the disease (32,33).
The association between DM and pain with the sex, race, and age group variables was described by (32). In our study, these variables were not associated with NP. However, we identified that other variables such as disease duration and glycemic variability could increase complications associated with pain, such as changes in temperature sensitivity, allodynia, amputations, ulcers, poor sleep quality, depression symptoms, changes in biomechanics, and worsening of quality of life, as revealed by other researchers (32-35).
Research aimed at identifying risk factors for developing NP has shown that its prevalence can increase dramatically when DM duration is longer than 20 years and is associated with smoking, high levels of fasting blood glucose and Hb1AC, low educational level, and underemployment (36). Our research revealed similar results, as many individuals in the sample were low-income rural workers with inadequate glycemic control.
It should be noted that the development of diseases caused by agricultural activity blurs NP screening, as it shares symptoms with several diseases of different etiologies. Degenerative changes in the spine and discopathies, which are very prevalent in the population, show symptoms such as pain, tingling, and sensitivity alterations like NP caused by DM, demonstrating the importance of NP-specific (35,37).
Studies carried out recently in Portugal point out to the prevalence of NP in T2DM (18-23 %) and its associations with the risk factors highlighted by DN4 pain screening. As in our study, increased prevalence of pain was related to high levels of Hb1aC, age group, and DM duration. There are no reports associating sex and ethnicity with pain (29,34).
In 2019, a Danish Center conducted a cohort survey of 5,514 people with diabetes, identifying an average of 4.6 years of disease duration and 10 % prevalence of pain through the MNSI and DN4 instruments. It reports significant associations between the appearance of NP and females, an increase in the age group, duration of the disease, BMI greater than 30, and smoking. The study also relates pain to allodynia, changes in temperature sensitivity, depression symptoms, and changes in the quality of sleep of individuals with an average DM duration of 12 years (32).
According to the 2019 Consensus on Diabetic Neuropathy, about 26 % of patients with T2DM developed chronic diabetic neuropathy, maintaining symptoms for more than three months. In fact, in our research, the main complications associated with NP in T2DM were the most referred to, i.e., allodynia, poor sleep quality, and night-time worsening of symptoms. These complications were present in more than 50 % of the people examined. Such manifestations are due to injury of the thin nerve fibers and a lack of glycemic control, producing changes in sensitivity, worsening burning and tingling symptoms, mainly at night, and influencing the quality of sleep of patients with painful diabetic neuropathy (37-39).
Pai et al. (40) investigated the risk factors for the onset of NP in Taiwan through an observational study with 2,837 people with T2DM screened using the MNSI and DN4 questionnaires. They found an NP prevalence of 21.2 %, related to advanced age, treatment with insulin, high blood pressure, high cholesterol, and Hb1Ac. These data are like those in the present study, in which the 1 % increase in Hb1Ac elevated three times the chance of developing NP (41).
High blood pressure, dyslipidemia, obesity, and smoking associated with painful diabetic neuropathy cause a higher prevalence of complications in individuals with more than ten years of disease, especially in more than half of people with NP. Current data show that this association occurs when 95.4 % of the studied individuals with pain have high blood pressure, 68.1 % dyslipidemia, 68.2 % obesity, and 54.5 % smoking, physically and emotionally impacting their quality of life (3, 38, 42).
The LANNS and DN4 instruments in patients with DN showed high scores for NP caused by nervous impairment of fine fibers Aõ and C, with symptoms such as hyperalgesia or allodynia. The BPI reported an average of seven points for pain intensity. These data corroborate an epidemiological study on the prevalence of painful diabetic neuropathy in the states of São Paulo, Ceará, and Bahia, Brazil, by applying the DN4 and BPI instruments, with scores above seven points (43). In conclusion, hyperalgesia and allodynia are related to high scores on the numerical pain scale, and this reflects in daily-life and professional activities, items assessed by the BPI (9).
Our results, associated with previously published studies, demonstrate that NP screening in diabetes mellitus can be performed using existing instruments validated in Portuguese. It is possible to design a systematic, integrative study to select instruments with more significant predictive value, sensitivity, and specificity and develop a more specific evidence-based protocol to screen and treat NP in T2DM. The sensitivity and specificity values of the instruments used in the screening were above 80 %, indicating good accuracy. Higher scores on the instruments were related to a longer duration of the disease, high levels of Hb1Ac, and insulin glycemic control (15,16,34).
It is possible to outline the profile of individuals with T2DM treated in Primary Health Care and their complications related to diabetic neuropathy and pain. For this, control measures, stricter risk factor assessments, and targeted programs are necessary to raise awareness of painful diabetic neuropathy in order to decrease or even prevent the resulting biomechanical and psychic changes.
Because diabetes is a disease with multisystem complications and pain is a multifactorial event, nursing and a multidisciplinary team are necessary to care for the patient holistically. Implementing a standardized protocol and monitoring the nursing and physiotherapy team for people with T2DM in the municipalities' primary health units is essential to maintain adequate glycemic control and, consequently, reduce the rates of NP complications and prevalence.
The present study had some limitations because the assessment of NP in people with diabetes mellitus relied on records made available by health units. Therefore, data from private institutions was not computed to obtain the actual prevalence of NP in the municipality.
Conclusion
NP screening allowed identifying pain in about 20 % of individuals surveyed, with moderate to severe intensity. NP was significantly associated with the use of alcohol and cigarettes, failure to have a balanced diet, and comorbidities such as obesity and high blood pressure. The probability of NP was three times greater upon a percentage increase in glycated hemoglobin. Complications such as changes in temperature sensitivity, allodynia, night-time worsening of symptoms, poor sleep patterns, depression symptoms, amputations, and ulcers were more frequent in patients with NP.
Pain is subjective, influenced by personal experiences and caused by various factors. It makes screening and treatment of painful diabetic neuropathy a challenge. However, due to the negative repercussions it can have on an individual's life, a highly accurate screening and assessment protocol and a multidisciplinary team led by nurses and physiotherapists are required to guide and treat patients more appropriately in Primary Health Care, especially those with known risk factors for developing NP.
Acknowledgment and financing
Thanks to the Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP) for awarding a grant to research process number 2017/26282-3.
Conflict of interest: None declared.
References
1. American Diabetes Association. Standards of Medical Care in Diabetes 2019. Diabetes Care. 2019;42(Suppl. 1):S4-S6. DOI: https://doi.org/10.2337/dc19-Srev01
2. Iqbal Z, Azmi S, Yadav R, Lim J, Malik RA, Alam U., et al. Diabetic Peripheral Neuropathy: Epidemiology, Diagnosis, and Pharmacotherapy. Clin Ther. 2018;40(6):828-849. DOI: https://doi.org/10.1016/j.clinthera.2018.04.001
3. Brazilian Diabetes Society. Diabetic Neuropathies: Position of the American Diabetes Association. 2019. DOI: https://doi.org/10.2337/dc16-2042
4. Sloan G, Shillo P, Selvarajah D, Wu J, Wilkinson ID, Tracey I, et al. A new look at painful diabetic neuropathy. Diabetes Res Clin Pract. 2018;144:177-191. DOI: https://doi.org/10.1016/j.diabres.2018.08.020
5. Shillo P, Sloan G, Greig M, Hunt L, Selvarajah D, Elliott J, et al. Painful and painless diabetic neuropathies: what is the difference? Curr Diabetes Rep. 2019;19:32. DOI: https://doi.org/10.1007/s11892-019-1150-5
6. Oliveira Volpe CM, Villar-Delfino PH, Ferreira dos Anjos PM, Nogueira-Machado JA. Cellular death, reactive oxygen species (ROS) and diabetic complications. Cell Death Dis. 2018:9,119. DOI: https://doi.org/10.1038/s41419-017-0135-z
7. Raputova J, Srotova I, Vlckova E, Sommer C, Uceyler N, Birklein F, et al. Sensory phenotype and risk factors for painful diabetic neuropathy: a cross-sectional observational study. Pain. 2017;158:2340-53. DOI: https://doi.org/10.1097/j.pain.0000000000001034
8. Brazilian Diabetes Society. Guidelines for the Brazilian Society of Diabetes: 2013-2014. São Paulo: Pharmaceutical Ac, 2014. Available from: https://nutritotal.com.br/pro/wp-content/uploads/sites/3/2014/04/342-diretrizessbd.pdf
9. Colloca L, Ludman T, Bouhassira D, Baron R, Dickenson AH, Yarnitsky D, et al. Neuropathic pain. Nat Rev Dis Primers. 2017;16(3). DOI: https://doi.org/10.1038/nrdp.2017.2
10. Raputova J, Vlckova E, Sommer C, Üçeyler N, Birklein F, Rittner HL, et al. Sensory phenotype and risk factors for painful diabetic neuropathy: a cross-sectional observational study. Pain. 2017;158(12):2340-2353. DOI: https://doi.org/10.1097/j.pain.0000000000001034
11. Sifuentes-Franco S, Padilla-Tejeda DE, Carrillo-Ibarra S, Miranda-Díaz AG. Oxidative Stress, Apoptosis, and Mitochon-drial Function in Diabetic Nephropathy. Int J Endocrinol. 2018;1. DOI: https://doi.org/10.1155/2018/1875870
12. Zhou W, Yuan Yao Y, Li J, Wu D, Zhao M, Yan Z, et al. TIGAR Attenuates High Glucose-Induced Neuronal Apoptosis via an Autophagy Pathway. Front Mol Neurosci. 2019;12:193. DOI: https://doi.org/10.3389/fnmol.2019.00193
13. Oliveira FB, Botelho KKP, Bezerra AR, Azevedo DIO, Couto CCS, Martins EF. Cross-cultural adaptation to Brazilian Portuguese of the Michigan neuropathy screening Instrument: MNSI - Brazil. Clinical Sales, Criteria and Tools. Arq Neuropsiquiatr. 2016;74(8):653-6612. DOI: https://doi.org/10.1590/0004-282X20160094
14. Barbosa M, Saavedra A, Severo M, Maier C, Carvalho D. Validation and reliability of the Portuguese version of the Michigan Neuropathy Screening Instrument. Pain Pract. 2016;17(4):514-521. DOI: https://doi.org/10.1111/papr.12479
15. Schestatsky C, Félix-Torres V, Chaves MLF, Câmara-Ehlers B, Mucenic T, Caumo W, et al. Brazilian Portuguese validation of the Leeds assessment of neuropathic symptoms and signs for patients with chronic pain. Pain Medicine,2011;12:1544-1550. DOI: https://doi.org/10.1111/j.1526-4637.2011.01221.x
16. Santos JG, Brito JO, De Andrade DC, Kaziyama VM, Ferreira KA, Souza I, et al. Translation to Portuguese and validation of the Douleur Neuropathique 4 questionnaire. J Pain. 2011;11(5):484-90. DOI: https://doi.org/10.1016/j.jpain.2009.09.014
17. Ferreira KA, Teixeira MJ, Mendonza TR, Cleeland CS. Validation of brief pain inventory to Brazilian patients with pain. Support Care Cancer. 2011;19(4):505-11. DOI: https://doi.org/10.1007/s00520-010-0844-7
18. Sarah A, Endrizzi MD, James P, Rathmell MD, Robert W, Hurley MD. Painful Peripheral Neuropathies. Essentials of Pain Medicine. 4ed. 2018: 273-282. DOI: https://doi.org/10.1016/B978-0-323-40196-8.00032-2
19. MNSI - Michigan Neuropathy Screening Instrument. Michigan Diabetes Research and Training Center. 2010. Available from: http://www.med.umich.edu/borc/profs/documents/svi/MNSI_patient.pdf
20. Sartor CD, Oliveira MD, Campos V, Ferreira JSSP, Sacco ICN. Cross-cultural adaptation and measurement properties of the Brazilian Version of the Michigan Neuropathy Screening Instrument. Braz J Phys Ther. 2018;22(3):222-230. DOI: https://doi.org/10.1016/j.bjpt.2017.10.004
21. Crucru G, Truini A. Neuropathic Pain: The Scope of the Problem. Pain and Therapy. 2017;6:1-3. DOI: https://doi.org/10.1007/s40122-017-0086-1
22. Carvalho AB, Garcia JS, Silva TKM, Riberio JVF. Translation and transcultural adaptation of pain quality assessment scale (PQAS) to Brazilian version. Braz J Anestesiol. 2016;66(1):94-104. DOI: https://doi.org/10.1016/j.bjane.2013.10.018
23. Agoons BB, Tchapmi D, Boli AO, Katte JC. Clinical utility of the DN4 questionnaire in the assessment of neuropathic pain in patients with type 2 diabetes: experience from a newly created diabetes clinic in Cameroon. PAMJ - Clinical Medicine. 2020:165(3). DOI: https://doi.org/10.11604/pamj-cm.2020.3.165.23789
24. Ministry of Health, Health Care Secretariat, Ordinance No. 1,083, of October 2, 2012. Available from: http://bvsms.saude.gov.br/bvs/saudelegis/sas/2012/prt1083_02_10_2012.html
25. Moreno RA, Moreno DH. Escalas de depressão de Montgomery & Asberg (MADRS) e de Hamilton (HAM-D). Rev Psiquiatr Clin. 1998;25:262-72. https://pesquisa.bvsalud.org/portal/resource/pt/lil-228053
26. Katigari MR, Ayatollahi H, Malek M, Haghighi MK. Fuzzy expert system for diagnosing diabetic neuropathy. World J Diabetes. 2017;8(2):80-88. DOI: https://doi.org/10.4239/wjd.v8.i2.80
27. Rouquayrol MZ, Gurge M. Epidemiologia e Saúde. 8 ed. Rio de Janeiro. Meedbook, 2017.
28. Feldman EL, Callaghan BC, Pop-Busui R, Zochodne DW, Wright DE, Bennett DL, et al. Diabetic neuropathy. Nat Rev Dis Primers. 2019;5(41). DOI: https://doi.org/10.1038/s41572-019-0092-1
29. Assunção A, Campos D, Marques R, Cunha I, Santos P, Martins A, et al. The impact of demographic and clinical characteristics on diabetic painful neuropathy. Rom J Intern Med. 2020;58(1):13-19. DOI: https://doi.org/10.2478/rjim-2019-0019
30. Hicks CW, Selvin E. Epidemiology of Peripheral Neuropathy and Lower Extremity Disease in Diabetes. Curr Diab Rep. 2019 Aug 27;19(10):86. DOI: https://doi.org/10.1007/s11892-019-1212-8
31. Cortez J, Reis C, Cardoso Y, Onofre A, Piovezan AP. Prevalência de dor neuropática e fatores associados em portadores de diabetes mellitus tipo 2 atendidos em ambulatório médico* SC. Rev Dor. 2014;15(4). DOI: https://doi.org/10.5935/1806-0013.20140055
32. Gylfadottir SS, Christensen DH, Nicolaisen SK, Andersen H, Callaghan BC, Itani M, Khan KS, et al. Diabetic polyneuro-pathy and pain, prevalence, and patient characteristics: a cross-sectional questionnaire study of 5,514 patients with recently diagnosed type 2 diabetes. Pain. 2019. DOI: https://doi.org/10.1097/j.pain.0000000000001744
33. Baron R, Maier C, Attal N, Binder A, Bouhassira D, Cruccu G, et al. Peripheral neuropathic pain: a mechanism-related organizing principle based on sensory profiles. Pain. 2017;158(2):261-272. DOI: https://doi.org/10.1097/j.pain.0000000000000753
34. Celik S, Yenidunya G, Temel E, Purisa S, Uzum AK, Gul N, et al. Utility of DN4 questionnaire in assessment of neuropathic pain and its clinical correlations in Turkish patients with diabetes mellitus. Kidney Care Diabetes. 2016;10(4): 259-64. DOI: https://doi.org/10.1016/j.pcd.2015.11.005
35. Jensen TS, Finnerup NB. Allodynia and hyperalgesia in neuropathic pain: clinical manifestations and mechanisms. Lancet Neurol. 2014;13(9):924-35. DOI: https://doi.org/10.1016/S1474-4422(14)70102-4
36. Garoushi S, Jonhson MI, Tashani OA. A cross-sectional study to estimate the point prevalence of painful diabetic neuropathy in Eastern Libya. BMC Public Health. 2019;19:78. DOI: https://doi.org/10.1186/s12889-018-6374-9
37. American Diabetes Association. Standards of Medical Care in Diabetes — 2017. Diabetes Care. 2017;40(1):S1-S135. https://care.diabetesjournals.org/content/diacare/suppl/2016/12/15/40.Supplement_1.DC1/DC_40_S1_final.pdf
38. Rolim LC, Koga da Silva EM, De Sá JR, Dib SA. A Systematic Review of Treatment of Painful Diabetic Neuropathy by Pain Phenotype versus Treatment Based on Medical Comorbidities. Front Neurol. 2017. DOI: https://doi.org/10.3389/fneur.2017.00285
39. Pop-Busui R, Boulton AJM, Feldman EL, Bril V, Freeman R, Malik RA, et al. Diabetic neuropathy: a position statement by the American Diabetes Association. Diabetes Care. 2017;40(1):136-54. DOI: https://doi.org/10.2337/dc16-2042
40. Pai YW, Lin CH, Lee IT, Chang MH. Prevalence and biochemical risk factors of diabetic peripheral neuropathy with or without neuropathic pain in Taiwanese adults with type 2 diabetes mellitus. Diabetes Metab Syndr. 2018;12(2):111-116. DOI: https://doi.org/10.1016/j.dsx.2017.09.013
41. Ponirakis G, Elhadd T, Chinnaiyan S, Dabbous Z, Siddiqui M, Al-Muhannadi H, et al. Prevalence and management of diabetic neuropathy in secondary care in Qatar. Diabetes Metab Res Rev. 2020;8. DOI: https://doi.org/10.1002/dmrr.3286
42. Pedrosa HC, Vilar L, Boulton AJM et al. Neuropatias e pés Diabéticos. ACF. 2013.
43. Udall M, Kudel I, Cappelleri JC, Sadosky A, King-Concialdi K, Parsons B, et al. Epidemiology of physician-diagnosed neuropathic pain in Brazil. J Pain Res. 2019;7(12):243-253. DOI: https://doi.org/10.2147/JPR.S160504